Lysosomes are sites of intracellular digestion and turnover of cellular components. Lysosomes (Gr. lysis, solution, + soma, body) are membrane-limited vesicles that contain a large variety of hydrolytic enzymes (more than 40) whose main function is intracytoplasmic digestion (Figures 2–24, 2–25, and 2–26). Lysosomes are particularly abundant in cells exhibiting phagocytic activity (eg, macrophages, neutrophilic leukocytes). Although the nature and activity of lysosomal enzymes vary depending on the cell type, the most common enzymes are acid phosphatase, ribonuclease, deoxyribonuclease, proteases, sulfatases, lipases, and beta-glucuronidase. As can be seen from this list, lysosomal enzymes are capable of breaking down most biological macromolecules. Lysosomal enzymes have optimal activity at an acidic pH.
Figure 2-24
Photomicrograph of a kidney tubule whose lumen appears in the center as a long slit. The numerous dark-stained cytoplasmic granules are lysosomes (L), organelles abundant in these kidney cells. The cell nuclei (N), some showing a nucleolus, are also seen in the photograph as dark-stained corpuscles. Toluidine blue stain. High magnification.
Figure 2-25
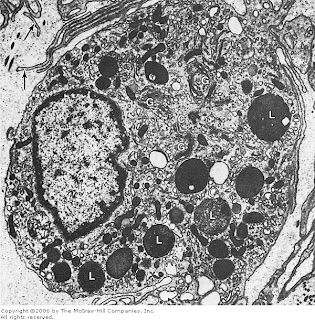
Electron micrograph of a macrophage. Note the abundant cytoplasmic extensions (arrows). In the center is a centriole (C) surrounded by Golgi cisternae (G). Secondary lysosomes (L) are abundant. x15,000.
Figure 2-26
Electron micrograph showing four dark secondary lysosomes surrounded by numerous mitochondria.
Lysosomes, which are usually spherical, range in diameter from 0.05 to 0.5 micrometer and present a uniformly granular, electron-dense appearance in electron micrographs. In a few cells, such as macrophages and neutrophilic leukocytes, primary lysosomes are larger, up to 0.5 micrometer in diameter, and thus are just visible with the light microscope.
The enveloping membrane separates the lytic enzymes from the cytoplasm, preventing the lysosomal enzymes from attacking and digesting cytoplasmic components. The fact that the lysosomal enzymes are practically inactive at the pH of the cytosol (~7.2) is an additional protection of the cell against leakage of lysosomal enzymes.
Lysosomal enzymes are synthesized and segregated in the RER and subsequently transferred to the Golgi complex, where the enzymes are modified and packaged as lysosomes. These enzymes have oligosaccharides attached to them with one or more of the mannose residues phosphorylated at the 6´ position by a phosphotransferase. There are receptors for mannose 6-phosphate-containing proteins in the RER and Golgi complex that allow these proteins to be diverted from the main secretory pathway and segregated in lysosomes.
Lysosomes that have not entered into a digestive event are identified as primary lysosomes.
Lysosomes can digest material taken into the cell from its environment. The material is taken into a phagosome or phagocytic vacuole (Figure 2–27); primary lysosomes then fuse with the membrane of the phagosome and empty their hydrolytic enzymes into the vacuole. Digestion follows, and the composite structure is now termed a secondary lysosome.
Figure 2-27
Current concepts of the functions of lysosomes. Synthesis occurs in the rough endoplasmic reticulum (RER), and the enzymes are packaged in the Golgi complex. Note the heterophagosomes, in which bacteria are being destroyed, and the autophagosomes, with RER and mitochondria in the process of digestion. Heterophagosomes and autophagosomes are secondary lysosomes. The result of their digestion can be excreted, but sometimes the secondary lysosome creates a residual body, containing remnants of undigested molecules. In some cells, such as osteoclasts, the lysosomal enzymes are secreted to the extracellular environment. Nu, nucleolus.
Secondary lysosomes are generally 0.2–2 micrometer in diameter and present a heterogeneous appearance in electron microscopes because of the wide variety of materials they may be digesting.
After digestion of the contents of the secondary lysosome, nutrients diffuse through the lysosomal-limiting membrane and enter the cytosol. Indigestible compounds are retained within the vacuoles, which are now called residual bodies (Figures 2–27 and 2–28). In some long-lived cells (eg, neurons, heart muscle), large quantities of residual bodies accumulate and are referred to as lipofuscin, or age pigment.
Figure 2-28
Section of a pancreatic acinar cell showing autophagosomes. Upper right: Two portions of the rough endoplasmic reticulum segregated by a membrane. Center: An autophagosome containing mitochondria (arrow) plus rough endoplasmic reticulum. Left: A residual body, with indigestible material. Arrowhead shows a cluster of coated vesicles.
Another function of lysosomes concerns the turnover of cytoplasmic organelles. Under certain conditions, a membrane may enclose organelles or portions of cytoplasm. Primary lysosomes fuse with this structure and initiate the lysis of the enclosed cytoplasm. The resulting secondary lysosomes are known as autophagosomes (Gr. autos, self, + phagein, to eat, + soma, body), indicating that their contents are intracellular in origin. Cytoplasmic digestion by autophagosomes is enhanced in secretory cells that have accumulated excess secretory product. The digested products of lysosomal hydrolysis are recycled by the cell to be reutilized by the cytoplasm.
Medical Apllication
In some cases, primary lysosomes release their contents extracellularly, and their enzymes act in the extracellular milieu. An example is the destruction of bone matrix by the collagenases synthesized and released by osteoclasts during normal bone tissue formation (see Chapter 8: Bone). Lysosomal enzymes acting in the extracellular milieu also play a significant role in the response to inflammation or injury. Several possible pathways relating to lysosome activities are schematically illustrated in Figure 2–27.
Lysosomes play an important role in the metabolism of several substances in the human body, and consequently many diseases have been ascribed to deficiencies of lysosomal enzymes. In metachromatic leukodystrophy, there is an intracellular accumulation of sulfated cerebrosides caused by lack of lysosomal sulfatases. In most of these diseases, a specific lysosomal enzyme is absent or inactive, and certain molecules (eg, glycogen, cerebrosides, gangliosides, sphingomyelin, glycosaminoglycans) are not digested. As a result, these substances accumulate in the cells, interfering with their normal functions. This diversity of affected cell types explains the variety of clinical symptoms observed in lysosomal diseases (Table 2–3).
| ||||||||||||||||||
| ||||||||||||||||||
I-cell disease (inclusion cell disease) is a rare inherited condition clinically characterized by defective physical growth and mental retardation and is due to a deficiency in a phosphorylating enzyme normally present in the Golgi complex. Lysosomal enzymes coming from the RER are not phosphorylated in the Golgi complex. Nonphosphorylated protein molecules are not separated to form lysosomes, instead following the main secretory pathway. The secreted lysosomal enzymes are present in the blood of patients with I-cell disease, whereas their lysosomes are empty. Cells of these patients show large inclusion granules that interfere with normal cellular metabolism.
| References
|





















39 comments:
Blog yang rame dengan pengunjung, sayang aku tak ngerti bahasanya :)
@Nurcahyaku2.co.cc ini rame gara2 pake traffic sob...hahaha... :D
lysosomes? lysosomes are found only in animals??
@Ladida that's true... :D
hohoo, it makes me confused, sometimes I think that lysosomes are found in plants :D
@Ladida memang betul...tapi kalo yang ditumbuhan namanya beda "peroksisom"...fungsinya sama aja...sbg penghasil enzim... :)
oooh, namanya aja yg beda, saya kira di tumbuhan ngga ada komponen sel kyk gini :D
di sekolah ngga ada atau saya yg kelewatan :P
ehmmm udah tau sih di SMA,tapi pas baca inggrisnya koq mumet ya??hahahaha
gw juga bingung nih bos...kayaknya banyak banget dah organel2 yang ada di sel...di setiap buku aja versinya ada yang beda2... T-T
Greetings I am so delighted I found your web site, I really found you by mistake, while
I was browsing on Askjeeve for something else, Nonetheless
I am here now and would just like to say cheers for a incredible post and a all round thrilling
blog (I also love the theme/design), I don’t have time to
look over it all at the minute but I have saved it and also added in your RSS
feeds, so when I have time I will be back to read much more, Please do keep up the
superb jo.
Feel free to visit my weblog - natural libido enhancers
i reckon both cheating and beating women is definitely bad,
just few things may started to mind when it comes to what type of
woman it might become. don't forget it will extremely much end up being the guy, not a great deal the woman. but i suppose if she seems less independent, like if she can't stand up for herself, or can't write for herself. or there could feel numerous issues that would make the guy cheat on her. but i really feel its much more the guy that would expect things like this, instead of the woman.
hope everythings alright for ya
Feel free to surf to my site ... how to attract women body language reading body language
Good post. I learn something totally new and challenging on blogs I stumbleupon everyday.
It will always be exciting to read through articles from other writers and use
a little something from other websites.
My website; stop grow review
You could definitely see your skills within the article you write.
The arena hopes for even more passionate writers such as you
who aren't afraid to say how they believe. All the time follow your heart.
Here is my weblog ... skinception demefface
Spectacular job with your website. Keep up because of the fabulous
get the job done.
Here is my web page: bad acne pictures
A fascinating discussion is worth comment. I think that you should write more on this subject, it might not be a taboo
matter but typically people don't discuss such topics. To the next! Many thanks!!
my web blog ... herbal libido enhancer
Just wish to say your article is as astounding. The clearness on your put up is just excellent and i can think you're an expert in this subject. Well with your permission allow me to snatch your feed to keep up to date with imminent post. Thanks 1,000,000 and please keep up the gratifying work.
My weblog :: eyelasticity reviews
Pretty nice post. I just stumbled upon your blog and wished to say that I've really enjoyed surfing around your blog posts. After all I'll be subscribing to your feed and
I hope you write again very soon!
my website women libido enhancement
Thank you for the good writeup. It actually used to be a
amusement account it. Look complex to more introduced
agreeable from you! However, how can we be in contact?
Feel free to visit my web site - hair removal
I was able to find good advice from your blog articles.
my page ... click through the up coming page
I really like your blog.. very nice colors & theme.
Did you make this website yourself or did you hire someone to do it for
you? Plz respond as I'm looking to create my own blog and would like to find out where u got this from. thanks
Here is my blog post; http://www.proman-help.isys-informatik.ch
Simply desired to assert I appricate the site. You have really invest a whole lot of energy into your
own article and it is truly just wonderfull!
Feel free to surf to my blog post :: my online income system
Thanks on your marvelous posting! I seriously enjoyed reading it, you can be a great author.
I will be sure to bookmark your blog and will come back at some point.
I want to encourage you continue your great work, have a nice morning!
My website ... lose body fat
You can definitely see your skills within the work you write.
The arena hopes for even more passionate writers such as you who are not afraid to say how they
believe. All the time go after your heart.
Here is my page; http://www.sbwire.com/press-releases/proactol-plus-review-new-studies-and-clinical-results-store-less-fat-and-lose-more-weight-238616.htm
I got this web site from my pal who told me on the topic of this website and at the moment this time I
am browsing this web site and reading very informative posts here.
Look into my page :: losing weight fast
people may possibly say ideology is dead and we reside
inside a post-ideological world, the problem with that is this sort?
of thinking is it really is self a form of ideology. so to acquire to your level
(effectively, to truly handle your level would take a lot of hours and ultimately have
to include way more than just my view), the issue is that the ideology that developed
the education method still exists and is getting widely purchased into.
it really is up to us to quit getting into the methods and ideologies that create
the problems.
my site attract women using body language
Just a rapid note to say numerous thanks to the tablets and also the diet
program. I started my diet plan 2 weeks back again at a weigh in 112.
4 Kilo and I'm now 104.4kg within just 13 times. Receiving excited about acquiring under 100KG after which on for the reduced 90s.
Also visit my web blog buy precision nutrition system
What a information of un-ambiguity and preserveness of valuable
experience concerning unpredicted emotions.
my web site - dermefface fx7 ()
Impressive job with your internet site. Keep up with the fabulous get the job done.
Feel free to surf to my blog; Sbwire.com
I am sure this piece of writing has touched all the internet viewers, its really really
fastidious article on building up new web site.
Here is my weblog ... building lean muscle
we simply like to allow you to know, we truly like the writing on your own website.
But I'm viewing it with my personal Google Chrome Browser and the display will not seem to be quite right. It's certainly not a big issue as I can still see and look over the posts but simply thought I would
let you know about that. brilliant operate. Cheers.
my blog http://www.sbwire.com/Press-releases/clearpores-review-clear-pores-acne-treatment-restore-your-body-Balance-latest-information-205445.htm
WOW just what I was searching for. Came here by searching for Romanian
My homepage - natural collagen production
Have you ever considered about including a little
bit more than just your articles? I mean, what you
say is valuable and all. Nevertheless think about if you added some great images
or videos to give your posts more, "pop"! Your content is
excellent but with images and video clips, this website could undeniably be one of the greatest in its
niche. Good blog!
Also visit my web site; body art
Hi, after reading this amazing piece of writing i am as well glad to share my experience here with colleagues.
Have a look at my web page ... music production lessons
My brother recommended I might like this blog.
He was entirely right. This post truly made my day.
You can not imagine just how much time I had spent for this information!
Thanks!
Also visit my web page :: huberg to open 2013 baltimore orioles season singing national anthem
Remarkable things here. I'm very happy to see your article. Thanks a lot and I'm looking forward to touch you.
Will you kindly drop me a e-mail?
my webpage - libido women late 30s
Hi there! Would you mind if I share your blog with my facebook group?
There's a lot of people that I think would really appreciate your content. Please let me know. Thanks
Feel free to visit my page; kyle leon somanabolic weight training
Wow, intriguing report many thanks! I'm going to possess to research this further and create a review on it for my weblog as well. I had never ever even heard about inexperienced coffee bean extract until now.
my web site ... weight loss smoothies mix
Create muscle very first. IIRC, the far more muscle
you have, the greater your resting metabolic process, never ever mind
your lively metabolism, along with the a lot more excess
fat you are going to then burn up.
My web-site mike chang monster mass diet
wow apa!!!!!!!!!!!!!!!!!!!!!!!!!!
Posting Komentar